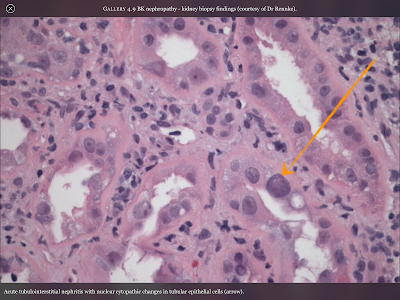
Pictured directly to the left is a cross-section of the the inside of a renal medulla (where filtration occurs). The small purple colored dots are the nuclei of the kidney’s epithelial cells which make up structures such as the walls of capillaries and microtubules. Because this is cross section the actual tubule is white space surrounded by a ring of cells, like looking at a pipe through the hole. The orange arrow is pointing at an epithelial cell that has been altered by the cytopathic effects of the BK Virus. It becomes enlarged and as you can see the tubule will begin to be closed up by the malformed cells, as well as the resulting scar-tissue.
 In the picture to the right, cells with a BK nuclear intrusion are highlighted using SV40 immunostaining. SV40 (Simian virus 40) is a virus in the same family as the BK (polyoma viruses) that can infect both humans and simians alike. Using it to stain locations of BK intrusion is another example of how we rely on research animals to be better able to help and understand ourselves, much like with rATG and hATG (see rATG post).
In the picture to the right, cells with a BK nuclear intrusion are highlighted using SV40 immunostaining. SV40 (Simian virus 40) is a virus in the same family as the BK (polyoma viruses) that can infect both humans and simians alike. Using it to stain locations of BK intrusion is another example of how we rely on research animals to be better able to help and understand ourselves, much like with rATG and hATG (see rATG post).
The final image shows fluorescent tagged immunoglobulin which is concentrated around areas of what is thought to be BK virus cytopathic manifestation.